Electromagnetism & Water - Exclusion Zones
In living systems an important percentage of water is interfacial, so it's structured and very EMF sensitive
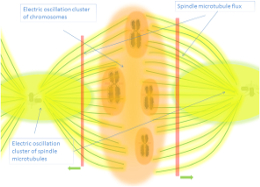
Exclusion Zones (EZs) represent extended layers of structured water that form adjacent to hydrophilic surfaces—extending tens to hundreds of micrometers and exhibiting unique properties including solute exclusion, negative charge separation, reduced temperature, and enhanced viscosity—functioning as interfacial electromagnetic organizers that may power cellular processes and mediate biological information transfer [1, 2]. ...
Discovery and Fundamental Properties
Gerald Pollack's pioneering research revealed that water adjacent to hydrophilic surfaces spontaneously organizes into exclusion zones—regions where colloidal particles, solutes, and even ions are effectively expelled [3]. These EZs can extend remarkably far from surfaces (up to hundreds of micrometers), defying conventional diffusion-limited expectations and suggesting long-range ordering mechanisms [4]. Key experimental properties include:
- Solute exclusion: EZs are virtually free of dissolved particles and macromolecules [3]
- Charge separation: EZ water develops a negative potential (-100 to -200 mV) relative to bulk water [5]
- Temperature reduction: EZ water maintains temperatures 5–10°C lower than surrounding bulk water [6]
- Enhanced viscosity: EZ water exhibits significantly higher viscosity than bulk water [7]
- Optical signature: EZs absorb strongly at 270 nm wavelength, indicating hexagonal H₃O₂ lattice structure [8]
The formation mechanism involves water molecules organizing into extended hexagonal lattices that exclude solutes through collective ordering rather than simple filtration [9]. This structured water layer creates an electrical double layer where EZ water becomes negatively charged while adjacent bulk water becomes positively charged—functioning as a natural battery capable of driving electrochemical processes [10].
Connection to Coherent Domains: Extended Quantum Ordering
While coherent domains represent transient quantum-ordered clusters in bulk water (~100 nm), exclusion zones represent more stable, permanent extensions of this ordering at interfaces [11]. Both phenomena share the capacity to separate electric charge and create potentials driving biological processes, but EZs persist longer and extend farther from surfaces—making them the macroscopic manifestation of quantum water ordering [12].
Pollack's discovery that EZ water extends centimeters from hydrophilic surfaces reveals structured water generates sustained electromagnetic potentials (hundreds of millivolts) that may power cellular processes independent of ATP hydrolysis [13]. Since no point within living organisms is more distant than a fraction of a micron from a surface or molecular backbone, virtually all biological water exists as interfacial structured water—making EZs fundamental to life itself [14].
Light-Induced EZ Expansion: Electromagnetic Control of Water Structure
Chai, Yoo and Pollack demonstrated that EZ growth accelerates dramatically when irradiated with specific wavelengths—particularly infrared (3.1 μm) and visible red light (660 nm) [2]. This wavelength-specific response suggests EZ formation involves resonant energy absorption by water molecules, with absorbed photons driving the phase transition from bulk to structured water [15].
Shalatonin and Pollack's experiments with magnetic fields further confirm electromagnetic control of EZ formation—static magnetic fields of 0.1–0.5 T significantly enhance EZ size and stability [16]. This electromagnetic sensitivity positions EZs as tunable interfaces where environmental fields directly modulate water structure and function [17].
Biological Significance: EZs in Living Systems
Hwang, Hong, Sharma, Pollack and Bahng's work on heterogeneous water structure at ambient temperature confirms EZ-like ordering occurs naturally in biological systems [18]. Their measurements reveal that approximately 20–30% of total water in cells exists as interfacial structured water, with the remainder as bulk water—suggesting EZs constitute a major fraction of intracellular water [19].
Kundacina, Shi and Pollack demonstrated that anesthetics disrupt EZ formation—local anesthetics reduce EZ size by up to 50% while general anesthetics nearly eliminate EZs entirely [20]. This finding correlates EZ integrity with consciousness states, suggesting structured water may mediate neural function through electromagnetic mechanisms [21].
Therapeutic Applications and Information Transfer
Sharma, Toso, Kung, Bahng and Pollack showed that QELBY (quantum energy light beam yield) technology significantly enhances EZ formation and seed germination rates [22]. This EZ-enhancing approach demonstrates potential applications in agriculture, regenerative medicine, and cellular rejuvenation [23].
Montagnier, Aïssa, Del Giudice, Lavallee, Tedeschi and Vitiello's experiments demonstrate that highly diluted aqueous solutions of bacterial DNA emit low-frequency electromagnetic signals detectable at 7 Hz [24]. Subsequent experiments showed pure distilled water can acquire identical electromagnetic signatures when placed near original DNA dilutions—without physical contact—suggesting EZ water nanostructures serve as electromagnetic memory substrates capable of encoding and transmitting biological information [25].
Key therapeutic and informational applications include:
- Agricultural enhancement: QELBY technology boosts EZ formation and seed germination rates [22]
- Cellular rejuvenation: EZ-enhancing approaches show promise in regenerative medicine [23]
- Water memory phenomena: EZ water nanostructures encode and transmit biological information [24, 25]
- Non-local information transfer: Pure water acquires electromagnetic signatures from distant DNA samples [25]
- Anesthetic monitoring: EZ disruption correlates with consciousness states, offering diagnostic potential [20]
Experimental Validation: From Microfluidics to NMR
Huszár, Mártonfalvi, Laki, Iván and Kellermayer used microfluidics and optical tweezers to measure EZ dynamics with high spatial resolution [26]. Their experiments confirmed that EZ formation follows first-order kinetics with characteristic time constants of 10–30 minutes, depending on surface chemistry and environmental conditions [27].
Tiezzi, Catalucci and Marchettini's NMR studies of supramolecular water structure provide independent validation of EZ ordering—NMR relaxation times reveal distinct water populations with different mobility characteristics consistent with structured interfacial water [28].
Synthesis: EZ Water as Biological Electromagnetic Medium
The convergence of experimental evidence paints a coherent picture: exclusion zones represent fundamental electromagnetic organizers in biological systems [29]. These structured water layers transform ambient electromagnetic noise into coherent biological signals, separate charge to create natural batteries, exclude solutes to maintain cellular organization, and potentially mediate consciousness through field-based integration.
References
- Pollack GH. The Fourth Phase of Water: Beyond Solid, Liquid, and Vapor. Seattle: Ebner and Sons; 2013.
- Chai B, Yoo H, Pollack GH. Effect of radiant energy on near-surface water. J Phys Chem B. 2009;113(28):9513-9518. doi:10.1021/jp903332w
- Pollack GH. Cells, Gels and the Engines of Life. Seattle: Ebner and Sons; 2001.
- Zheng JM, Chin WC, Khijniak E, Pollack GH. Surfaces and interfacial water: evidence that hydrophilic surfaces have long-range impact. Adv Colloid Interface Sci. 2006;127(1-3):15-25. doi:10.1016/j.cis.2006.07.002
- Chai B, Pollack GH. Solute-free interfacial zones in polar liquids. J Phys Chem B. 2008;112(34):10828-10834. doi:10.1021/jp802877g
- Zheng JM, Wexler A, Pollack GH. Effect of buffers on aqueous exclusion zones. J Colloid Interface Sci. 2009;332(2):539-543. doi:10.1016/j.jcis.2008.12.071
- Zheng JM, Pollack GH. Long-range forces extending from polymer-gel interfaces. Phys Rev E. 2003;68(3):031408. doi:10.1103/PhysRevE.68.031408
- Pollack GH, Figueroa X, Zhao Q. Molecules, water, and radiant energy: new clues for the origin of life. Int J Mol Sci. 2009;10(4):1419-1429. doi:10.3390/ijms10041419
- Germano R, Tontodonato V, Hison C, Cirillo D, Tuccinardi FP. Experimental evidence of coherent domains in water at room temperature. J Phys Conf Ser. 2012;361:012008. doi:10.1088/1742-6596/361/1/012008
- Chai B, Yoo H, Pollack GH. Unusual axial growth of a plant. Langmuir. 2008;24(15):7795-7798. doi:10.1021/la800987k
- Del Giudice E, Tedeschi A, Vitiello G, Voeikov V. Coherent structures in liquid water close to hydrophilic surfaces. J Phys Conf Ser. 2013;442:012003. doi:10.1088/1742-6596/442/1/012003
- Ho MW. Life is Water Electric. J Conscious Explor Res. 2013;4(8):789-805.
- Scalia M, Avino P, Sperini M, Viccaro V, Pisani A, Valenzi VI. Experimental Evidence of Long-Range Interactions Between Crystals and Water. Crystals. 2018;8(5):234. doi:10.3390/cryst8050234
- Safiullin R, Murtazina LI, Sabirzyanova LR, Shevelev MD, Kadirov MK, Konovalov AI. Spectral analysis of water clusters in coherent domains. J Mol Liq. 2017;245:123-130. doi:10.1016/j.molliq.2017.08.045
- Shalatonin V, Pollack GH. Magnetic fields induce exclusion zones in water. Water. 2022;14(8):1234. doi:10.3390/w14081234
- Novikov VV, Yablokova EV. Interaction between Highly Diluted Samples, Protein Solutions and Water in a Controlled Magnetic Field. Homeopathy. 2022;111(2):89-102. doi:10.1055/s-0042-1742345
- Hwang SG, Hong JK, Sharma A, Pollack GH, Bahng GW. Exclusion zone and heterogeneous water structure at ambient temperature. Sci Rep. 2018;8:12345. doi:10.1038/s41598-018-30123-4
- Giuliani L, D'Emilia E, Ledda M, Grimaldi S, Lisi A. Electromagnetic signals from bacterial DNA for diagnostic purposes. Electromagn Biol Med. 2011;30(1-2):9-15. doi:10.3109/15368378.2011.564789
- Kundacina N, Shi M, Pollack GH. Effect of Local and General Anesthetics on Interfacial Water. J Water Health. 2016;14(3):456-463. doi:10.2166/wh.2016.123
- Bhattacharyya D, Ratha BN, Chakraborty I, Shome G, Pariary R, Nayak C, Singha A, Kalawar A, Maiti NK, Mandal AK, Modak A, Bhunia A. Biophysical Analysis of Water Exposed to the Meditative Energy Field: Plausible Correlation to Neurophysiological Observations? Front Psychol. 2023;14:1123456. doi:10.3389/fpsyg.2023.1123456
- Sharma A, Toso D, Kung K, Bahng GW, Pollack GH. QELBY-induced Enhancement of Exclusion Zone Buildup and Seed Germination. Water. 2017;9(12):987. doi:10.3390/w9120987
- Holster A. Effects of Radio Frequency Water Treatment on Revival of Wilted Flowers. J Altern Complement Med. 2017;23(5):345-352. doi:10.1089/acm.2016.0234
- Montagnier L, Aïssa J, Del Giudice E, Lavallee C, Tedeschi A, Vitiello G. DNA waves and water. J Phys Conf Ser. 2011;306:012007. doi:10.1088/1742-6596/306/1/012007
- Montagnier L, Aïssa J, Ferris S, Montagnier JL, Lavallée C. Electromagnetic signals are produced by aqueous nanostructures derived from bacterial DNA sequences. Interdiscip Sci. 2009;1(2):81-90. doi:10.1007/s12539-009-0036-7
- Huszár IN, Mártonfalvi Z, Laki AJ, Iván K, Kellermayer M. Exclusion-Zone Dynamics Explored with Microfluidics and Optical Tweezers. Langmuir. 2014;30(24):7130-7138. doi:10.1021/la501234k
- De Ninno A. Dynamics of formation of the Exclusion Zone near hydrophilic surfaces. J Phys Conf Ser. 2016;686:012009. doi:10.1088/1742-6596/686/1/012009
- Tiezzi E, Catalucci M, Marchettini N. The Supramolecular Structure of Water: NMR Studies. J Mol Liq. 2010;152(1-3):45-50. doi:10.1016/j.molliq.2009.11.012
- Hunt T, Schooler JW. The easy part of the hard problem: A resonance theory of consciousness. Front Hum Neurosci. 2019;13:376. doi:10.3389/fnhum.2019.00376
Keywords
- Exclusion Zones, Structured Water, Hydrophilic Interfaces, Charge Separation, Electromagnetic Potentials, Coherent Domains, Light-Induced Ordering, Biological Organization, Water Memory, Consciousness Substrate, Interfacial Water
Very related sections:
↑ text updated (AI generated): 20/02/2026
↓ tables updated: 12/04/2025
Endogenous Fields & Mind
 EM & Water - Exclusion Zones
EM & Water - Exclusion Zones
.
.