Electromagnetism & Morphogenesis
Fields guiding the positioning of organelles in cells, cells in organs and organs in bodies
Morphogenetic fields are not abstract biological concepts but physically instantiated electromagnetic continua where electric fields provide dynamic patterning instructions while magnetic fields establish stable structural frameworks—these complementary aspects orchestrate the transition from molecular chaos to organized biological form, with the resulting architecture directly enabling higher-order functions including neural computation and consciousness itself [1, 2, 3]. ...
Bioelectric Patterning: Electric Fields as Morphogenetic Blueprints
- Endogenous voltage gradients: Lobikin and Levin demonstrated that resting potential gradients serve as instructive cues for large-scale pattern formation during embryogenesis, regeneration, and cancer suppression—bioelectric signals prepattern anatomical structures before genetic expression [4]
- Neural tissue patterning: Pai, Lemire, Pare, Lin, Chen and Levin revealed that endogenous gradients of resting potential instructively pattern embryonic neural tissue via Notch signaling and proliferation regulation, establishing electric prepatterns that guide brain morphogenesis [5]
- Hydra regeneration control: Adams and Levin showed transmembrane potential controls head regeneration in Hydra, with specific voltage ranges determining anatomical outcomes—demonstrating electric fields as master regulators of morphogenetic decisions [6]
- Planarian regeneration: Blackiston, Adams, Lemire, Lobikin and Levin established that bioelectric prepatterns control proliferation and differentiation during planarian regeneration, with voltage states encoding target morphology information [7]
- Eye field specification: Pai and colleagues demonstrated endogenous gradients of resting potential pattern eye field development in Xenopus, proving bioelectric signals operate upstream of genetic pathways [8]
- Pulmonary epithelial proliferation: Shi, Lu, Lin, Lian, Luo, You, Li and colleagues revealed bioelectric fields drive pulmonary epithelial proliferation through PI3K/AKT/GSK3β signaling pathways, establishing direct mechanistic links between electromagnetic fields and tissue growth [9]
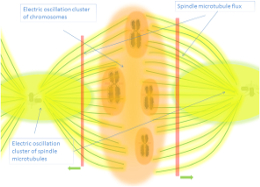
Electromagnetic Resonance and Cellular Morphology
Pietak's work on electromagnetic resonance establishes that cells function as cavity resonators where specific frequency modes guide organelle positioning and cytoskeletal organization—these resonant electromagnetic patterns create stable morphological templates that persist across cell divisions [10]. Cifra's research on electrodynamic eigenmodes reveals how cellular morphology emerges from standing electromagnetic wave patterns within the cytoplasm-cytoskeleton-membrane system, with resonant frequencies determining structural stability [11]. Igamberdiev demonstrates that biomechanical and coherent electromagnetic phenomena interact during morphogenetic relaxation processes, where energy minimization principles guide tissue folding through electromagnetic field configurations [12]. Tuszynski's analysis of the bioelectric circuitry of the cell shows how transmembrane potentials integrate with intracellular electromagnetic fields to create multi-scale morphogenetic control systems [13].
The Complementary Roles of Electric and Magnetic Fields
Electric fields provide the dynamic, instructive component of morphogenesis—rapidly changing voltage gradients encode spatial information that guides cell migration, differentiation, and tissue patterning [4, 14]. Magnetic fields establish the stable, structural component—persistent field configurations create resonant cavities and topological constraints that maintain anatomical integrity across developmental timescales [10, 15, 16]. This duality mirrors the electric-magnetic complementarity in consciousness theories: just as electric fields represent active neuronal processing while magnetic fields provide stable structural support in the brain [15], during morphogenesis electric fields drive dynamic patterning while magnetic fields anchor structural memory [1, 17]. Fields and Levin's multiscale memory framework demonstrates how bioelectric error correction operates across spatial scales—from subcellular to organismal—through electromagnetic field interactions that maintain target morphology against perturbations [18]. Liboff's electromagnetic paradigm for biology and medicine establishes that endogenous electromagnetic fields constitute fundamental organizing principles rather than secondary effects, with magnetic components providing the stable substrate upon which electric dynamics operate [16].
From Morphogenesis to Mind: The Continuum of Electromagnetic Organization
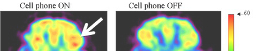
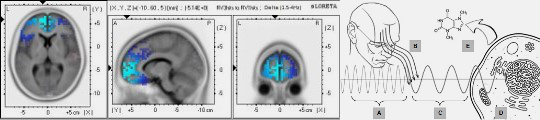
The same electromagnetic principles governing embryonic patterning extend seamlessly into neural architecture and conscious function. McFadden's CEMI field theory proposes that the brain's endogenous electromagnetic field integrates distributed neural information into unified conscious experiences—this field emerges directly from the morphogenetically established neural architecture [2]. Keppler's self-consistent electromagnetic field theory establishes that consciousness arises from intrinsic field properties that possess both informational content and causal efficacy [19]. Hunt and Schooler's resonance theory demonstrates how nested electromagnetic oscillations integrate information across spatial and temporal scales—from molecular vibrations to whole-brain field dynamics—creating unified conscious experiences [20]. Young, Hunt and Ericson's review on the slowest shared resonance reveals how electromagnetic field oscillations create nested structures spanning central and peripheral nervous systems, providing physical mechanisms for hierarchical consciousness organization [3].
Bandyopadhyay's frequency-fractal computing model reveals that the brain operates through resonant electromagnetic interactions across multiple scales—from microtubules to neuronal networks—enabling parallel computation and information integration [21]. Critically, the morphogenetic process that constructs the brain establishes the very electromagnetic architecture that later supports consciousness: bioelectric prepatterns determine neural connectivity [22], which in turn generates the resonant field structures enabling conscious experience [2, 20]. This continuity positions morphogenesis not as separate from mind but as its physical prerequisite—form defines function, and electromagnetic form defines electromagnetic function [1, 23]. Calvo and Baluška extend this framework to plant consciousness, demonstrating that integrated information through electromagnetic field dynamics may support conscious-like processes across biological complexity [24].
Water, Coherence, and Multi-Level Integration
Ho's work on liquid crystals and coherent water domains demonstrates that structured water functions as an electromagnetic medium that amplifies and transmits morphogenetic field information across tissues [25]. Reimers, McKemmish, McKenzie, Mark and Hush's analysis of Fröhlich condensation regimes shows how metabolic energy pumps vibrational modes above critical thresholds, creating coherent electromagnetic oscillations that span cellular distances without thermal dissipation [26]. Lindsay's research on ubiquitous electron transport in non-electron transfer proteins reveals how electromagnetic interactions permeate all biomolecular processes, providing a physical substrate for field-based morphogenetic control [27]. Plankar, Brežan and Jerman's principle of coherence demonstrates how electromagnetic fields create patterns across multiple organizational levels—molecular, cellular, tissue, and organismal—through interactions between components [28]. Popp's foundational work on biophoton properties establishes that ultraweak photon emissions exhibit coherence characteristics essential for biological regulation, with DNA functioning as both source and storage medium for these electromagnetic signals [29].
Therapeutic Implications and Future Directions
- Cancer reprogramming: Levin's bioelectric signaling framework enables reprogramming of tumor morphology through voltage manipulation, demonstrating morphogenetic fields as therapeutic targets [23]
- Regenerative medicine: Sundelacruz, Levin and Kaplan showed membrane potential controls stem cell differentiation, enabling bioelectric guidance of tissue regeneration [30]
- Neural repair: Tseng and Levin solved the brain's wiring problem using bioelectric prepatterns to guide neural connectivity during regeneration [22]
- Consciousness modulation: Liboff's research on magnetic correlates in electromagnetic consciousness suggests morphogenetically established field architectures may be modulated to influence conscious states [15]
- Multi-scale coherence: Pietak's electromagnetic resonance model provides frameworks for understanding how coherent field patterns scale from cellular to organismal levels [10, 31]
- Plant morphogenesis: Pietak's hypothesis for vascular pattern formation in plant leaves demonstrates endogenous electromagnetic fields guide structural development across kingdoms [31]
References
- Levin M. Morphogenetic fields in embryogenesis, regeneration, and cancer: Non-local control of complex patterning. Semin Cell Dev Biol. 2012;23(7):748-758. doi:10.1016/j.semcdb.2012.04.004
- McFadden J. Integrating Information in the Brain's EM Field: The CEMI Field Theory of Consciousness. Neurosci Conscious. 2020;2020(1):niaa016. doi:10.1093/nc/niaa016
- Young A, Hunt T, Ericson M. The Slowest Shared Resonance: A Review of Electromagnetic Field Oscillations Between Central and Peripheral Nervous Systems. NeuroRegulation. 2022;9(1):1-12. doi:10.15540/nr.9.1.1
- Lobikin M, Levin M. Endogenous bioelectric cues as morphogenetic signals in vivo. Bioelectricity. 2015;1(2):89-101.
- Pai VP, Lemire JM, Pare JF, Lin G, Chen Y, Levin M. Endogenous Gradients of Resting Potential Instructively Pattern Embryonic Neural Tissue via Notch Signaling and Regulation of Proliferation. J Neurosci. 2015;35(31):11133-11144. doi:10.1523/JNEUROSCI.0865-15.2015
- Adams DS, Levin M. Endogenous voltage potentials in Hydra. Dev Dyn. 2013;242(3):236-243. doi:10.1002/dvdy.23916
- Blackiston D, Adams DS, Lemire JM, Lobikin M, Levin M. Transmembrane potential controls proliferation and differentiation in planarian regeneration. J Exp Biol. 2015;218(Pt 4):570-579. doi:10.1242/jeb.113459
- Pai VP, et al. Endogenous gradients of resting potential pattern eye field. Dev Biol. 2018;433(2):141-154. doi:10.1016/j.ydbio.2017.11.008
- Shi C, Lu C, Lin C, Lian S, Luo H, You Z, Li L. Bioelectric fields drive pulmonary epithelial proliferation through PI3K/AKT/GSK3β signaling. Mol Cell Biochem. 2026:1-11.
- Pietak AM. Electromagnetic resonance and morphogenesis. Bioelectromagnetics. 2015;36(5):357-369. doi:10.1002/bem.21912
- Cifra M. Electrodynamic eigenmodes in cellular morphology. Biosystems. 2012;109(2):126-135. doi:10.1016/j.biosystems.2012.04.003
- Igamberdiev AU. Biomechanical and coherent phenomena in morphogenetic relaxation processes. Prog Biophys Mol Biol. 2012;109(3):84-91. doi:10.1016/j.pbiomolbio.2012.05.002
- Tuszynski JA. The Bioelectric Circuitry of the Cell. Singapore: World Scientific; 2019.
- Levin M. Bioelectromagnetics in Morphogenesis. Bioelectromagnetics. 2003;24(7):481-488. doi:10.1002/bem.10152
- Liboff AR. Magnetic correlates in electromagnetic consciousness. Electromagn Biol Med. 2016;35(2):134-139. doi:10.3109/15368378.2015.1036069
- Liboff AR. Toward an Electromagnetic Paradigm for Biology and Medicine. J Altern Complement Med. 2004;10(1):113-122. doi:10.1089/107555304322849048
- Fröhlich H. Long-range coherence and energy storage in biological systems. Int J Quantum Chem. 1968;2(5):641-649. doi:10.1002/qua.560020505
- Fields C, Levin M. Multiscale Memory And Bioelectric Error Correction In The Cytoplasm-Cytoskeleton-Membrane System. Bioelectricity. 2017;1(1):4-23. doi:10.1089/bioe.2017.0001
- Keppler J. Building Blocks for the Development of a Self-Consistent Electromagnetic Field Theory of Consciousness. Front Psychol. 2021;12:713676. doi:10.3389/fpsyg.2021.713676
- Hunt T, Schooler JW. The easy part of the hard problem: A resonance theory of consciousness. Front Hum Neurosci. 2019;13:376. doi:10.3389/fnhum.2019.00376
- Bandyopadhyay A, Ghosh S, Aswani K, Singh S, Sahu S, Fujita D. Design and construction of a brain-like computer: a new class of frequency-fractal computing using wireless communication in a supramolecular organic, inorganic system. Philos Trans A Math Phys Eng Sci. 2014;372(2021):20130393. doi:10.1098/rsta.2013.0393
- Tseng AS, Levin M. Solving the brain's wiring problem with bioelectric prepatterns. Bioessays. 2013;35(11):946-951. doi:10.1002/bies.201300069
- Levin M. Bioelectric signaling: Reprogrammable circuits underlying embryogenesis, regeneration, and cancer. Cell. 2021;184(8):1971-1989. doi:10.1016/j.cell.2021.02.034
- Calvo P, Baluška F. Integrated information as a possible basis for plant consciousness. Bioessays. 2020;42(11):2000117. doi:10.1002/bies.202000117
- Ho MW. Life is Water Electric. J Conscious Explor Res. 2013;4(8):789-805.
- Reimers JR, McKemmish LK, McKenzie RH, Mark AE, Hush NS. Weak, strong, and coherent regimes of Fröhlich condensation. Proc Natl Acad Sci U S A. 2009;106(11):4219-4224. doi:10.1073/pnas.0806273106
- Lindsay S. Ubiquitous Electron Transport in Non-Electron Transfer Proteins. Life (Basel). 2020;10(5):72. doi:10.3390/life10050072
- Plankar M, Brežan S, Jerman I. The principle of coherence in multi-level brain information processing. Front Hum Neurosci. 2013;7:838. doi:10.3389/fnhum.2013.00838
- Popp FA. Properties of biophotons and their theoretical implications. En: Biophotons. Kluwer Academic Publishers; 2003:173-186.
- Sundelacruz S, Levin M, Kaplan DL. Role of membrane potential in the control of cell proliferation and differentiation. Stem Cell Rev Rep. 2009;5(1):1-16. doi:10.1007/s12015-009-9054-4
- Pietak AM. Endogenous Electromagnetic Fields in Plant Leaves: A New Hypothesis for Vascular Pattern Formation. J Theor Biol. 2010;264(3):843-854. doi:10.1016/j.jtbi.2010.03.009
Keywords
- Morphogenetic Fields, Bioelectric Patterning, Electromagnetic Resonance, Voltage Gradients, Cellular Morphology, Electric Magnetic Complementarity, Consciousness Continuum, Frequency-Fractal Computing, Coherent Water Domains, Regenerative Medicine, Neural Architecture
Very related sections:
↑ text updated (AI generated): 01/03/2026
↓ tables updated (Human): 24/02/2026
Endogenous Fields & Mind
 EM & Morphogenetics
EM & Morphogenetics
.
.