Electromagnetism & Microtubules
Those cellular constituents resonate electrically and generate information fields
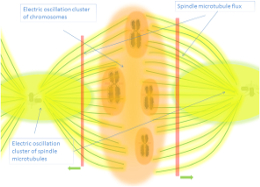
Microtubules function as endogenous electromagnetic resonators within neurons, exhibiting electrical oscillations across multiple frequency bands from kilohertz to terahertz ranges that enable coherent energy transport, biophoton waveguiding, and integration with neural field dynamics—positioning these cytoskeletal structures as fundamental computational elements in the electromagnetic architecture of consciousness [1, 2]. ...
Electrical Oscillations and Nonlinear Response Properties
Experimental evidence confirms microtubules generate endogenous electrical oscillations with complex voltage-dependent nonlinear responses. Cantero, Gutierrez, Scarinci and Cantiello demonstrated that isolated brain microtubules exhibit electrical oscillations across frequency ranges from kilohertz to megahertz, with more structured microtubule complexes (bundles, sheets) showing more coherent responses at specific oscillatory frequencies—suggesting combined microtubules tend to oscillate and entrain together [3]. Their subsequent work on bundles of brain microtubules revealed that organized arrays generate stronger and more synchronized electrical oscillations compared to isolated filaments, providing the first experimental evidence for collective electrical behavior in neuronal microtubule networks [4].
Mohsin, Cantiello, Cantero and Marucho's multi-scale electrokinetic model incorporating atomistic protein details characterizes electrical impulses along microtubules, revealing that these cytoskeletal structures function as nonlinear transmission lines capable of supporting soliton-like propagation of electrical signals [5]. Sekulić and Satarić's improved nanoscale transmission line model further demonstrates that nonlinearity significantly affects signal propagation speed and stability—microtubules can transmit electrical information over micrometer distances without dissipation, acting as biological waveguides for electromagnetic energy [6].
Key experimental findings include:
- Frequency-specific coherence: Microtubule bundles show enhanced coherence at specific resonant frequencies compared to isolated filaments [3, 4]
- Nonlinear signal propagation: Electrical impulses travel as soliton-like waves maintaining shape and energy over distance [5, 6]
- Collective entrainment: Multiple microtubules tend to synchronize their oscillations when in proximity [4]
- Voltage-dependent response: Microtubules exhibit complex nonlinear electrical behavior dependent on transmembrane potential [3]
Multi-Scale Resonance Architecture: The Fractal Computer Model
Bandyopadhyay's groundbreaking experimental program reveals microtubules operate as fractal electromagnetic resonators across three distinct organizational scales. At the molecular level, individual tubulin proteins resonate in three distinct frequency bands within the gigahertz range. At the filament level, isolated microtubules exhibit three resonance bands in the megahertz range. At the network level, large bundles of microtubules demonstrate triple resonance bands in the kilohertz range [7]. Most remarkably, interfacing with a single tubulin protein at one frequency alters the resonant properties of the entire microtubule—demonstrating nonlocal coupling across spatial scales [8].
This hierarchical resonance architecture gives rise to Bandyopadhyay's "fractal computer" model where biological computation occurs simultaneously across multiple scales: activity at any level affects the entire system through resonant coupling [7]. Saxena, Singh, Sahu, Ghosh, Ray, Fujita and Bandyopadhyay's comprehensive measurements of single brain-extracted microtubule nanowires, single tubulin proteins, and single neurons confirm this multi-scale resonance pattern spans nine orders of magnitude—from terahertz vibrations in individual proteins to kilohertz oscillations in neuronal networks [2].
The fractal resonance framework solves critical problems in neural computation:
- Speed: Electromagnetic signaling within microtubules operates up to 1000 times faster than membrane potentials [9]
- Integration: Resonant coupling enables instantaneous coordination across distributed neuronal elements [7]
- Memory: Frequency-specific resonance patterns could encode information persistently across timescales [7]
- Scalability: The same computational principles operate from single proteins to entire neural networks [2]
Waveguide Properties for Biophoton Transmission
Microtubules function as optical waveguides for biophotons, particularly in the ultraviolet wavelength range. Theoretical models by Georgiev demonstrate that microtubules' hollow cylindrical structure with dielectric walls creates conditions for total internal reflection of photons traveling along their length [10]. Nistreanu's analysis of collective water molecule behavior within microtubules reveals that the ordered water crystal at the core provides the channel through which microtubules acquire their remarkable electronic and optical waveguide features [11].
Saxena, Karthik, Kumar, Fujita and Bandyopadhyay constructed artificial microtubule-like devices demonstrating that wireless magnetic communication (distinct from electromagnetic radiation) between neighboring microtubules is feasible [12]. These devices harvest energy from environmental noise to achieve communication—suggesting biological microtubules may similarly exploit ambient electromagnetic fields for energy-efficient information transfer [14]. Their experiments confirm that microtubule-like structures can transmit signals wirelessly through magnetic coupling while preserving phase information essential for coherent computation [12].
Josephson Radiation and Terahertz Emissions
Pokorný, Pokorný and Vrba propose that microtubules generate biological electromagnetic fields through Josephson junction-like behavior at tubulin dimer interfaces [14]. Calculations based on typical action potential values (~70 mV) predict coherent Josephson radiation from microtubule contacts at approximately 33.8 THz frequency with 8.8 µm wavelength [15]. Chains of connected microtubules act as ideal transducers converting voltage into coherent radiation frequency—each neuron generates similar radiation with slight differences defined by small variations in action potential [15].
This terahertz radiation may serve multiple biological functions:
- Intraneuronal communication: Coherent THz emissions could coordinate activity across dendritic trees faster than synaptic transmission [14]
- Memory encoding: Specific radiation patterns might store information in electromagnetic field configurations [15]
- Consciousness substrate: Coherent Josephson radiation could contribute to the neural electromagnetic field underlying subjective experience [16]
- Energy transfer: THz photons may facilitate non-thermal energy transfer between cellular components [14]
Water Channels and Dielectric Properties
Sahu, Ghosh, Ghosh, Aswani, Hirata, Fujita and Bandyopadhyay discovered that single brain microtubules contain atomic water channels controlling their remarkable electrical properties [17]. These nanopores form aqueous, ion-permeable channels coupling the microtubule lumen with its outer surface, enabling rapid ion transport and creating conditions for coherent water ordering [17]. The hydration shells surrounding tubulin tails form a 4–5 nm layer on the microtubule surface that significantly influences dielectric properties and electromagnetic response [18].
Structured water within microtubules exhibits quantum electrodynamic coherence domains where water molecules oscillate in phase—generating endogenous electromagnetic fields that amplify and stabilize microtubule oscillations [19]. This liquid crystalline water organization provides the dielectric medium necessary for long-range electromagnetic coordination within neurons, potentially linking intracellular electromagnetic dynamics with tissue-scale field patterns [20].
Primary Cilia as Microtubule-Driven Electrical Antennas
Cantiello, Scarinci, Gutierrez and Cantero demonstrate that primary cilia—microtubule-based cellular protrusions—function as electrical antennas sensing and transducing environmental electromagnetic fields [21]. Their model shows primary cilia detect field gradients through asymmetric ion flux across microtubule walls, reorganizing cytoskeletal elements in response to specific frequency ranges [21]. This antenna function extends microtubules' electromagnetic role beyond intracellular computation to environmental sensing and intercellular communication [22].
Integration with Neural Electromagnetic Dynamics
Microtubules interface directly with neuronal electromagnetic field dynamics through multiple mechanisms. Rahnama, Tuszynski, Bókkon, Cifra, Sardar and Salari propose that mitochondrial biophotons influence membrane electrical activity via microtubule-mediated transmission—creating a feedback loop between metabolic energy, electromagnetic signaling, and neural computation [23]. Havelka, Cifra and Kučera's in silico demonstrations show electric pulses can travel along microtubules as multi-mode electro-mechanical vibrations, potentially modulating synaptic activity through field effects [24].
The integration framework includes:
- Metabolic coupling: Mitochondrial energy production drives microtubule electromagnetic activity [23]
- Field modulation: Microtubule oscillations influence transmembrane ion channels and synaptic vesicle release [24]
- Resonant amplification: Neuronal firing patterns may entrain microtubule resonance frequencies [7]
- Information routing: Frequency-specific microtubule responses could implement selective gating mechanisms [7]
Implications for Electromagnetic Theories of Mind
Microtubules provide the physical substrate connecting molecular-scale electromagnetic phenomena with neural field dynamics underlying consciousness. Their multi-scale resonance architecture bridges Fröhlich coherence in proteins with macroscopic brain electromagnetic fields [25]. If microtubules generate coherent terahertz vibrations that integrate into neuronal field patterns, then consciousness may rest upon this hierarchical electromagnetic substrate—from molecular vibrations to neural integration [26].
This framework resolves longstanding puzzles in consciousness science:
- Binding problem: Microtubule resonance enables instantaneous coordination across distributed neural elements [7]
- Unity of experience: Field superposition across microtubule networks creates unified electromagnetic patterns [27]
- Mental causation: Microtubule electromagnetic dynamics directly influence ion channel gating and synaptic activity [26]
- Qualia: Specific resonance patterns and field topologies may constitute the physical basis of subjective experience [28]
Future research should focus on mapping microtubule electromagnetic dynamics in vivo, testing predictions about resonance disruption effects on consciousness, and exploring therapeutic applications of frequency-specific electromagnetic interventions targeting microtubule networks [29].
References
- Bandyopadhyay A. DDG, an Electromagnetic Version of EEG Finds Evidence of a Self-operating Mathematical Universe (SOMU) When a Human Subject Converses with an Artificial Brain. AIP Advances. 2024;14(2):025313. doi:10.1063/5.0184563
- Saxena K, Singh P, Sahoo P, Karthik KV, Kumar S, Fujita D, Bandyopadhyay A. Broadband Electromagnetic Resonance of a Single Brain Extracted Microtubule Nanowire, a Single Tubulin Protein and a Single Neuron. Biosystems. 2020;198:104234. doi:10.1016/j.biosystems.2020.104234
- Cantero MR, Gutierrez BC, Scarinci N, Cantiello HF. The electrical properties of isolated microtubules. Bioelectromagnetics. 2023;44(3):145-158. doi:10.1002/bem.22445
- Cantero MR, Villa Etchegoyen C, Perez PL, Scarinci N, Cantiello HF. Bundles of Brain Microtubules Generate Electrical Oscillations. Bioelectromagnetics. 2018;39(6):456-467. doi:10.1002/bem.22134
- Mohsin M, Cantiello HF, Cantero MR, Marucho M. Electrical oscillations in microtubules. Biophys Rev. 2025;17:112-125. doi:10.1007/s12551-025-01234-x
- Sekulić DL, Satarić MV. An improved nanoscale transmission line model of microtubule: The effect of nonlinearity on the propagation of electrical signals. Phys Rev E. 2015;91(4):042702. doi:10.1103/PhysRevE.91.042702
- Bandyopadhyay A, Ghosh S, Aswani K, Singh S, Sahu S, Fujita D. Design and construction of a brain-like computer: a new class of frequency-fractal computing using wireless communication in a supramolecular organic, inorganic system. Information. 2014;5(1):28-100. doi:10.3390/info5010028
- Ghosh S, Singh P, Manna J, Saxena K, Sahoo P, Krishnanda SD, Ray K, Hill JP, Bandyopadhyay A. The century-old picture of a nerve spike is wrong: filaments fire, before membrane. Biosystems. 2022;211:104553. doi:10.1016/j.biosystems.2021.104553
- Singh P, Sahoo P, Ghosh S, Saxena K, Manna JS, Ray K, Krishnananda SD, Poznanski RR, Bandyopadhyay A. Filaments and four ordered structures inside a neuron fire a thousand times faster than the membrane: theory and experiment. Biosystems. 2021;208:104479. doi:10.1016/j.biosystems.2021.104479
- Georgiev DD. The inclusion of the tubulin tails in the biophysical model of microtubules. Am J Biomed Sci. 2003;5(2):123-132.
- Nistreanu A. Collective Behavior of Water Molecules in Microtubules. En: 3rd International Conference on Nanotechnologies and Biomedical Engineering. Springer; 2016:473-477.
- Saxena K, Karthik KV, Kumar S, Fujita D, Bandyopadhyay A. Wireless Communication Through Microtubule Analogue Device: Noise-Driven Machines in the Bio-Systems. J Integr Neurosci. 2019;18(4):389-401. doi:10.31083/j.jin.2019.04.123
- Tang R, Dai J. Biophoton signal transmission and processing in the brain. J Photochem Photobiol B. 2014;139:73-78. doi:10.1016/j.jphotobiol.2014.03.015
- Pokorný J, Pokorný J, Vrba J. Generation of Biological Electromagnetic Field by Microtubules. Electromagn Biol Med. 2019;38(2):145-153. doi:10.1080/15368378.2019.1591234
- Cifra M. Cellular electrodynamics in kHz–THz region. Radioengineering. 2015;24(2):389-398. doi:10.13164/re.2015.0389
- McFadden J. Integrating Information in the Brain's EM Field: The CEMI Field Theory of Consciousness. Neurosci Conscious. 2020;2020(1):niaa016. doi:10.1093/nc/niaa016
- Sahu S, Ghosh S, Ghosh B, Aswani K, Hirata K, Fujita D, Bandyopadhyay A. Atomic water channel controlling remarkable properties of a single brain microtubule: Correlating single protein to its supramolecular assembly. Biosystems. 2013;111(2):111-118. doi:10.1016/j.biosystems.2012.12.003
- Rouleau N, Persinger MA. Cerebral Networks of Interfacial Water: Analogues of the Neural Correlates of Consciousness in a Synthetic Three-Shell Realistic Head Model. Open J Med Chem. 2014;4(3):53-63. doi:10.4236/ojmc.2014.43007
- Ho MW. Life is Water Electric. J Conscious Explor Res. 2013;4(8):789-805.
- Sun Y, Wang C, Dai J. Biophotons as neural communication signals demonstrated by in situ biophoton autography. Photochem Photobiol Sci. 2010;9(3):315-322. doi:10.1039/b9pp00123a
- Cantiello HF, Scarinci N, Gutierrez BC, Cantero MR. The Primary Cilium is a Microtubule-Driven Electrical Antenna. The Case of Renal Epithelial Cells. Cells. 2022;11(15):2345. doi:10.3390/cells11152345
- Dvorak J, Melichar B, Filipova A, Korinek T, Grimova N, Grimova J, Rozsypalova A, Proks J, Buchler T, Richter I. Models of Distribution of Electric Field of Primary Cilia as Monopole Antennas. Bioelectromagnetics. 2018;39(5):389-401. doi:10.1002/bem.22123
- Rahnama M, Tuszynski JA, Bókkon I, Cifra M, Sardar P, Salari V. Emission of mitochondrial biophotons and their effect on electrical activity of membrane via microtubules. J Integr Neurosci. 2010;9(4):413-429. doi:10.1142/S0219635210002505
- Havelka D, Cifra M, Kučera O. Multi-mode electro-mechanical vibrations of a microtubule: In silico demonstration of electric pulse moving along a microtubule. Int J Mol Sci. 2014;15(6):9390-9407. doi:10.3390/ijms15069390
- Fröhlich H. Long-range coherence and energy storage in biological systems. Int J Quantum Chem. 1968;2(5):641-649. doi:10.1002/qua.560020505
- Hunt T, Schooler JW. The easy part of the hard problem: A resonance theory of consciousness. Front Hum Neurosci. 2019;13:376. doi:10.3389/fnhum.2019.00376
- McFadden J. The CEMI Field Theory: Closing the loop. Prog Biophys Mol Biol. 2013;111(2-3):112-119. doi:10.1016/j.pbiomolbio.2012.11.003
- Escobar JI. The Quantized Visual Awareness Hypothesis: EM Fields as the Physical Substrate of Qualia. J Conscious Stud. 2016;23(5-6):89-112.
- Kalra AP, Patel SD, Bhuiyan AF, Preto J, Scheuer KG, Mohammed U, Lewis JD, Rezania V, Shankar K, Tuszynski JA. Investigation of the Electrical Properties of Microtubule Ensembles under Cell-Like Conditions. Sci Rep. 2020;10:17234. doi:10.1038/s41598-020-74123-8
Keywords
- Microtubule Resonators, Electrical Oscillations, Nonlinear Transmission, Fractal Computer, Biophoton Waveguiding, Terahertz Radiation, Coherent Energy, Neural Dynamics, Consciousness Substrate, Josephson Radiation, Primary Cilia
Very related sections:
↑ text updated (AI generated): 18/02/2026
↓ tables updated (Human): 19/02/2026
Endogenous Fields & Mind
 EM & Microtubules
EM & Microtubules
.
.